Scientists at the Medical Research Council (MRC) Epidemiology Unit at the University of Cambridge in collaboration with Huma Therapeutics Limited have published results from the Fenland COVID-19 study, showing that participants had high, sustained levels of engagement with a smartphone app developed by Huma to collect information including health related measurements from individual participants.
The COVID-19 pandemic accelerated interest in the use of digital and mobile health solutions for remote health monitoring. But while these technologies have shown benefits for patients with chronic or acute health conditions, less is known about their utility in population-based health research, where it is becoming increasingly hard to recruit participants and keep them engaged over months or even years.
The results, published in the Journal of Medical Internet Research, involved 62.61% (2524/4031) of the participants in the Fenland COVID-19 study, who were recruited from the longitudinal Fenland study, making it the largest population-based study to-date exploring how digital technologies can support population research. The Fenland COVID-19 study examined the potential of identifying people who are infected in the pre-symptomatic phase using information collected from individuals remotely, and also aimed to measure the effect of the national COVID-19 restrictions including social distancing on health-related behaviours such as diet, physical activity, wellbeing and mental health.
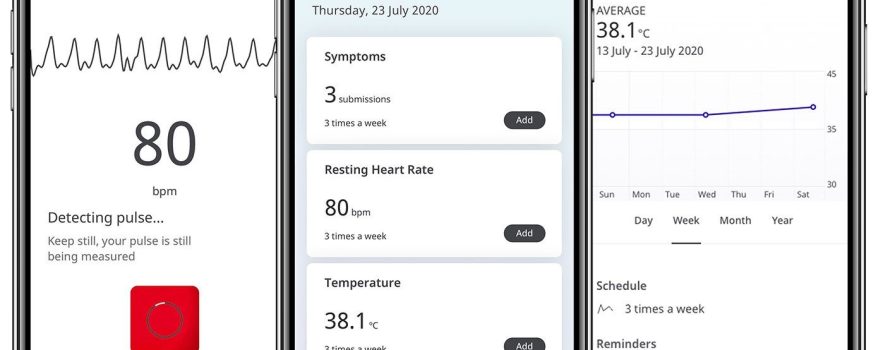
From the participants who were invited to register to use the app, 90.21% (2277/2524) completed the app-based onboarding process and signed e-consent. In addition to using the study app, which was available for both iPhone and Android, each was also sent a digital pulse oximeter to measure blood oxygen levels, and a thermometer. They were provided with remote set-up assistance and were asked to record the following biomarkers:
- blood oxygen saturation (three times per week)
- body temperature (three times per week)
- resting heart rate (three times per week)
- activity levels (measured passively)
- respond to monthly questionnaires
- blood spot samples to test for the presence of coronavirus antibodies.
Participants had a positive experience with the study app, finding it easy to use and quick to report measurements and symptoms. They took part in the observational study for at least 6 months and most kept completing measures until asked to stop; there was minimal drop off in engagement over the study period. On average, people used the study app for 34.5 weeks (7.9 months), with only 2.5% of participants withdrawing from the study. It was interesting to see a higher engagement rate amongst the participants aged over 65.
Dr Kirsten Rennie, a Senior Research Associate at MRC Epidemiology Unit and an expert in quantitative measures of physical activity and diet, who led the study said:
Enrolment and retention in traditional cohort-based observational studies is a constant challenge and participation has been declining in recent years. Here we saw not only great enrolment and retention, but also engagement which has helped us create a useful checklist for other researchers to follow.”
Dr Arrash Yassaee, Global Clinical Director at Huma, said:
Huma is committed to building robust clinical and scientific evidence for its technology. The high level of engagement and retention we’ve seen in this study is very encouraging. User-friendly clinical trial technologies such as Huma’s have great potential to transform population-based health research by increasing access and reducing the burden on participants. This kind of data collection is incredibly valuable for understanding health and disease processes in the real world and gathering insights that can make a difference to people’s lives.”
Checklist for mobile health (mHealth) engagement in population-based studies:
- Ensure the remote measurement technology platform is compatible for both iOS and Android, and compatible with devices at least 5-years old
- Make the initial engagement as simple as possible with minimum steps to complete
- To reduce digital exclusion, produce both written and video instructions on how to download the app and complete enrolment in the study – test these with users
- Be clear with participants on the commitment of taking part: communicate the length of study and level of engagement required
- If feasible for the study design, establish a routine for active data collection, for example, specific days of week
- Avoid active data questions for participants where the default or most common answer is an absence or null response e.g. reporting no symptoms
- Consider passive data collection options where appropriate (i.e. data collection that does not involve participants entering measurements)
- Collect quantitative and qualitative data to monitor participant engagement
Read more
- Rennie K.L., Lawlor E.R., Yassaee A., Booth A., Westgate K., Sharp S.J., Tyrrell C.S., Aral M., Wareham N.J. Engagement with mHealth COVID-19 digital biomarker measurements in a longitudinal cohort study: a mixed methods evaluation. J Med Internet Res. 2022 Oct 3. doi: 10.2196/40602. Epub ahead of print. PMID: 36194866.

 MRC Epidemiology Unit
MRC Epidemiology Unit